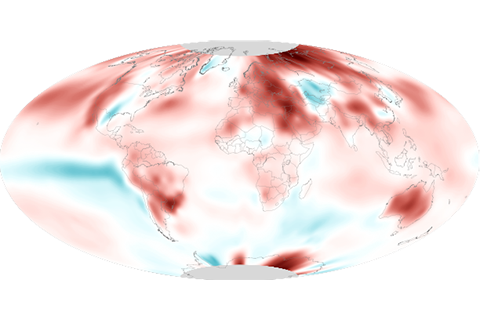
September 2020 was the hottest September on record for the globe, continuing a sweltering year.
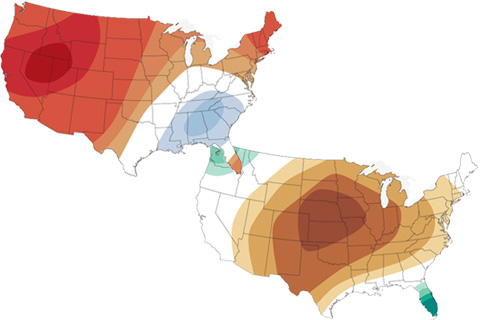
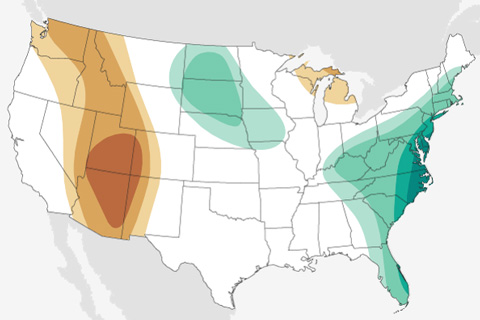
The latest maps from NOAA's Climate Prediction Center show where October 2020 is forecast to be much hotter and drier than average in the U.S. It's not a great outlook for the wildfire-affected West.
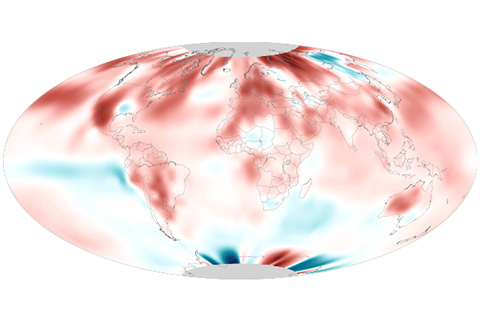
August 2020 was the second-hottest August on record, and it ended the hottest summer on record for the Northern Hemisphere.
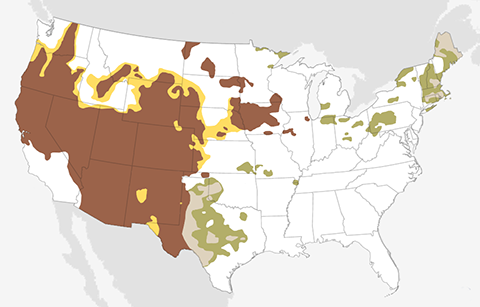
The September 2020 temperature and precipitation outlook favors a warmer- and drier-than-average September across the western United States, and a wetter-than-average month across the south-central Plains and much of the East.
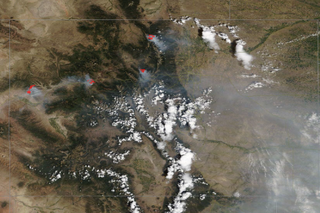
A Colorado summer: Drought, wildfires and smoke in 2020
August 20, 2020
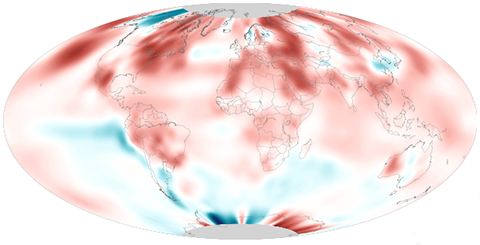
July 2020 was the second-warmest July on record for the globe, as 2020 continues its scorching path to one of the hottest years on record.

Join three heat experts to talk about how we map, monitor, and lessen the impacts of urban heat islands.
The August 2020 outlook favors hotter-than-average temperatures along both coasts, while tropical moisture is likely to lead to a wetter-than-average August along the East Coast.
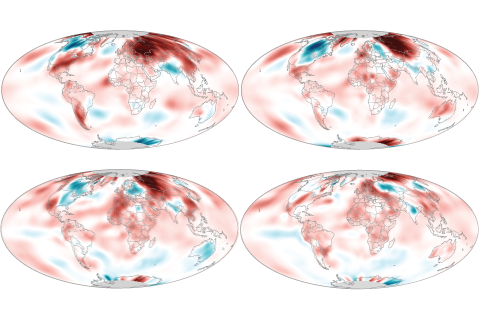
June 2020 broke the streak of each month being warmest or second warmest on record by being only the third-warmest June on record.
